Discover Paper Talk
Paper Talk

607 Episodes
Reverse
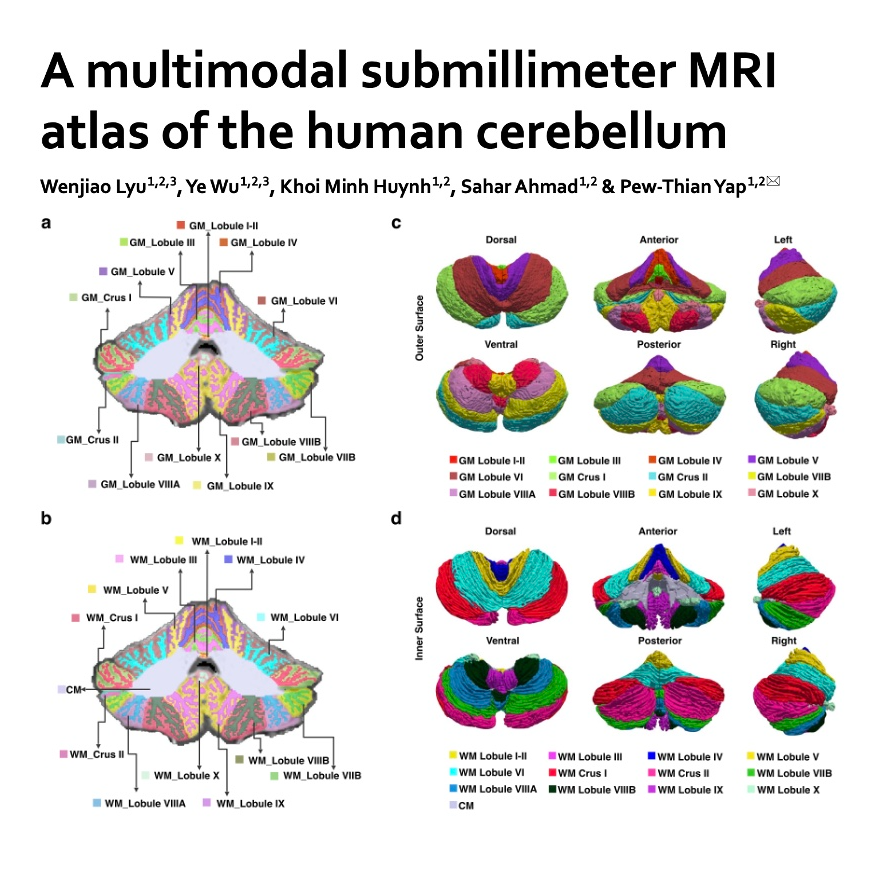
Researchers have developed a high-resolution multimodal MRI atlas to better understand the human cerebellum, a complex brain structure vital for motor coordination and cognitive processes. Unlike previous models that simplified its anatomy, this new tool utilizes submillimeter imaging to provide a detailed view of gray and white matter across 24 distinct regions per hemisphere. By incorporating microstructure maps and fiber tractography, the atlas illustrates how the cerebellum connects to various parts of the cerebrum, including areas responsible for memory, attention, and language. The study highlights that specific zones like Crus I and Crus II are linked to higher-order functions, showing different levels of myelination compared to motor regions. Ultimately, this comprehensive resource offers a new framework for investigating how the "little brain" influences neurodevelopment and brain disorders.References: Lyu W, Wu Y, Huynh K M, et al. A multimodal submillimeter MRI atlas of the human cerebellum[J]. Scientific reports, 2024, 14(1): 5622.
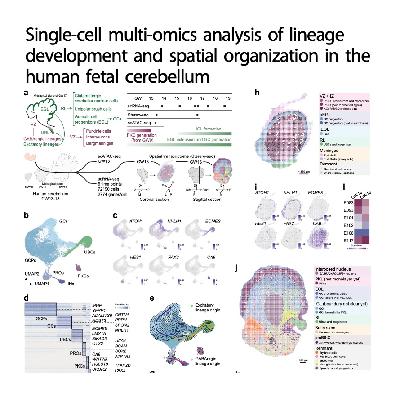
Researchers in this study utilized advanced multi-omics techniques to create a detailed map of the human fetal cerebellum between gestational weeks 13 and 18. By integrating single-cell RNA sequencing, chromatin analysis, and spatial transcriptomics, they successfully identified the distinct genetic programs that drive cellular diversity. The team discovered a unique group of neuroepithelium at the rhombic lip and characterized various neuronal subtypes, including Purkinje and unipolar brush cells. Their work highlights how gene regulatory networks control the transformation of progenitors into mature neurons within specific layers of the brain. Additionally, the study provides a cross-species comparison that reveals how certain gene expression patterns differ between humans and mice. These findings offer a high-resolution view of cerebellar development and provide a valuable framework for understanding the origins of motor and cognitive circuits.References: Yang F, Zhao Z, Zhang D, et al. Single-cell multi-omics analysis of lineage development and spatial organization in the human fetal cerebellum[J]. Cell Discovery, 2024, 10(1): 22.
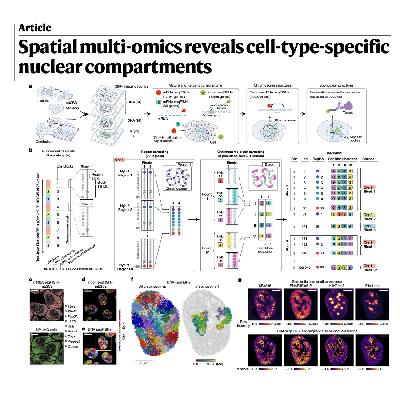
The paper details the development of two-layer DNA seqFISH+, a high-resolution imaging technology designed to map the three-dimensional organization of the genome in single cells. By simultaneously analyzing over 100,000 genomic loci, the nascent transcriptome, and various subnuclear structures, researchers can observe how chromatin compartments vary across different cell types in complex tissues like the mouse cerebellum. The study reveals that active chromatin regions remain relatively consistent, while repressive regions—specifically those marked by H3K27me3 and H4K20me3—are highly cell-type specific. These repressive structures dictate radial chromosomal positioning and inter-chromosomal interactions, particularly distinguishing neurons from glial cells. Ultimately, this multi-omics approach provides a detailed view of how nuclear architecture influences gene regulation and spatial genome folding in native biological contexts.References: Takei Y, Yang Y, White J, et al. Spatial multi-omics reveals cell-type-specific nuclear compartments[J]. Nature, 2025, 641(8064): 1037-1047.
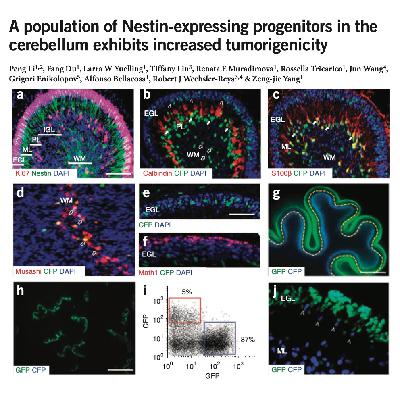
The paper describes the discovery of Nestin-expressing progenitors (NEPs), a previously unidentified type of cell in the developing cerebellum. While Nestin is typically a marker for multi-use stem cells, researchers found that these specific NEPs are committed solely to becoming granule neurons. These cells are distinct from the more common granule neuron precursors (GNPs) because they reside in a different layer of the cerebellum and remain quiescent rather than rapidly dividing. Crucially, NEPs lack robust DNA repair mechanisms, making them highly susceptible to genetic errors. The study demonstrates that when certain signaling pathways are overactive, these cells are more likely than GNPs to undergo genomic instability. Consequently, NEPs appear to be a primary cell of origin for medulloblastoma, a common type of brain tumor.References: Li P, Du F, Yuelling L W, et al. A population of Nestin-expressing progenitors in the cerebellum exhibits increased tumorigenicity[J]. Nature neuroscience, 2013, 16(12): 1737-1744.
This research reveals that the human cerebellum is organized by a multifaceted gradient that coordinates structural and functional development from childhood to adulthood. By analyzing large MRI datasets and postmortem tissue, scientists discovered that microstructural properties, such as myelin density, vary across cerebellar lobules in a pattern that mirrors their functional roles in sensory and cognitive tasks. This anatomical hierarchy is not static; rather, lobules associated with higher-level thinking show prolonged growth and significant tissue proliferation compared to primary sensory regions. Quantitative imaging and proteomic analysis confirmed these findings, showing that the cerebellum is far more structurally diverse and dynamic than previously assumed. Furthermore, the study demonstrates that functional signals within the cerebellum are tightly coupled with these physical changes, providing a new framework for identifying atypical development in disorders like autism. Overall, the evidence suggests that the cerebellum’s growth-rate gradient is influenced by both genetic factors and the organ's unique evolutionary history in humans.References: Liu X, d’Oleire Uquillas F, Viaene A N, et al. A multifaceted gradient in human cerebellum of structural and functional development[J]. Nature neuroscience, 2022, 25(9): 1129-1133.
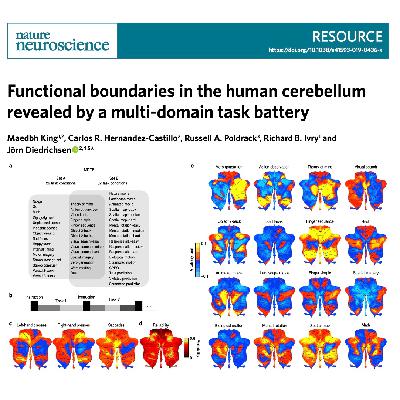
This research presents a comprehensive functional map of the human cerebellum, moving beyond its traditional role in motor control to identify its involvement in complex cognitive and emotional processes. By utilizing a multi-domain task battery (MDTB) with 47 unique conditions, the authors developed a novel parcellation of the cerebellar cortex into ten distinct functional regions. Their findings demonstrate that traditional anatomical lobular boundaries fail to represent functional divisions, whereas their new task-based atlas significantly improves the prediction of brain activity. The study further reveals that while the cerebellum contains discrete functional subregions, the fine-grained details of these patterns are largely unique to each individual. Ultimately, this work provides a new functional framework for future neuroimaging research and a deeper understanding of the cerebellum's diverse contributions to human behavior.References: King M, Hernandez-Castillo C R, Poldrack R A, et al. Functional boundaries in the human cerebellum revealed by a multi-domain task battery[J]. Nature neuroscience, 2019, 22(8): 1371-1378.
This research article examines the functional development of the human cerebellum from birth through age five using over 1,000 high-quality brain scans. The study reveals that the cerebellum is already connected to higher-order cognitive networks at birth, with these interactions strengthening as children mature. By mapping the brain's functional topography, the authors demonstrate that cerebellar organization evolves from diffuse patterns in infancy to more focal, adult-like structures by age three. The findings also highlight significant functional asymmetry and sex-specific developmental trajectories, particularly in networks related to language and executive control. Ultimately, this work provides a foundational map of typical cerebellocortical connectivity, offering critical insights for future research into early-onset neurodevelopmental disorders.References: Lyu W, Thung K H, Huynh K M, et al. Functional development of the human cerebellum from birth to age five[J]. Nature Communications, 2025, 16(1): 6350.
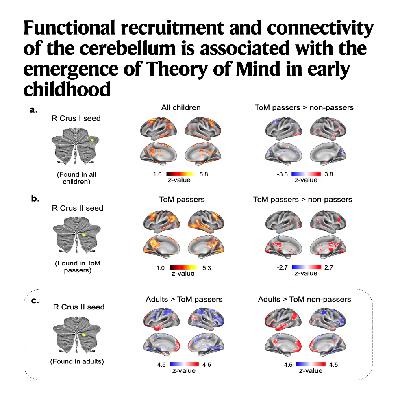
This research study investigates how the cerebellum contributes to the development of Theory of Mind (ToM) in children. By analyzing fMRI data from children and adults, the authors discovered that the Crus I and II regions of the cerebellum are specifically activated when children begin to understand false beliefs. The study highlights a significant shift in functional connectivity, where the cerebellum primarily sends information upstream to the cerebral cortex in childhood, while the direction is reversed in adulthood. These findings suggest that the cerebellum is essential for establishing the neural architecture required for social cognition early in life. Consequently, the research helps explain why early cerebellar injuries can lead to more profound and lasting social deficits than those sustained later in development.References: Manoli A, Van Overwalle F, Grosse Wiesmann C, et al. Functional recruitment and connectivity of the cerebellum is associated with the emergence of Theory of Mind in early childhood[J]. Nature communications, 2025, 16(1): 5273.
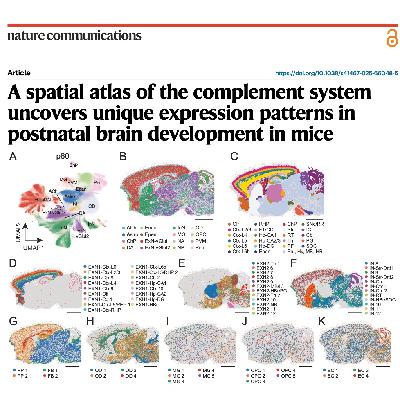
This research article presents a comprehensive spatial transcriptomic atlas of the complement system within the mouse brain, spanning from embryonic stages to adulthood. Using MERFISH technology, the authors mapped the expression of 51 complement-related genes, discovering that most are produced locally with high cellular and regional diversity. While the classical pathway increases during maturation to facilitate synaptic pruning, the study identifies a previously unknown role for the alternative pathway in early brain development. Specifically, the researchers found that Masp3, an alternative pathway activator, is vital for brain maturation and proper cortical neuron distribution. Their findings demonstrate that a lack of Masp3 leads to spatial memory deficits and significant systemic growth issues. Overall, the study highlights how the brain maintains a unique immune environment by strictly regulating complement activity to prevent inflammatory damage.References: Zhang Y, Watson B, Rattan A, et al. A spatial atlas of the complement system uncovers unique expression patterns in postnatal brain development in mice[J]. Nature Communications, 2025.
This article from Nature Neuroscience critically evaluates Lesion Network Mapping (LNM), a popular neuroimaging technique used to link brain injuries to psychiatric and neurological disorders. The authors highlight a major procedural flaw where diverse conditions like addiction, depression, and epilepsy appear to map onto nearly identical brain circuits. Their mathematical analysis reveals that LNM fundamentally simplifies into a repetitive sampling of the same normative connectivity data, regardless of the specific disease being studied. Consequently, the resulting maps frequently reflect nonspecific properties of the brain's "degree" or overall connectivity rather than unique biological markers. This discovery suggests that many published findings may be methodological artifacts rather than distinct discovery of disorder-specific networks. Ultimately, the study serves as a foundational caution for the neuroscience community, urging the development of more precise analytical tools.References: van den Heuvel M P, Libedinsky I, Quiroz Monnens S, et al. Investigating the methodological foundation of lesion network mapping[J]. Nature Neuroscience, 2026: 1-11.
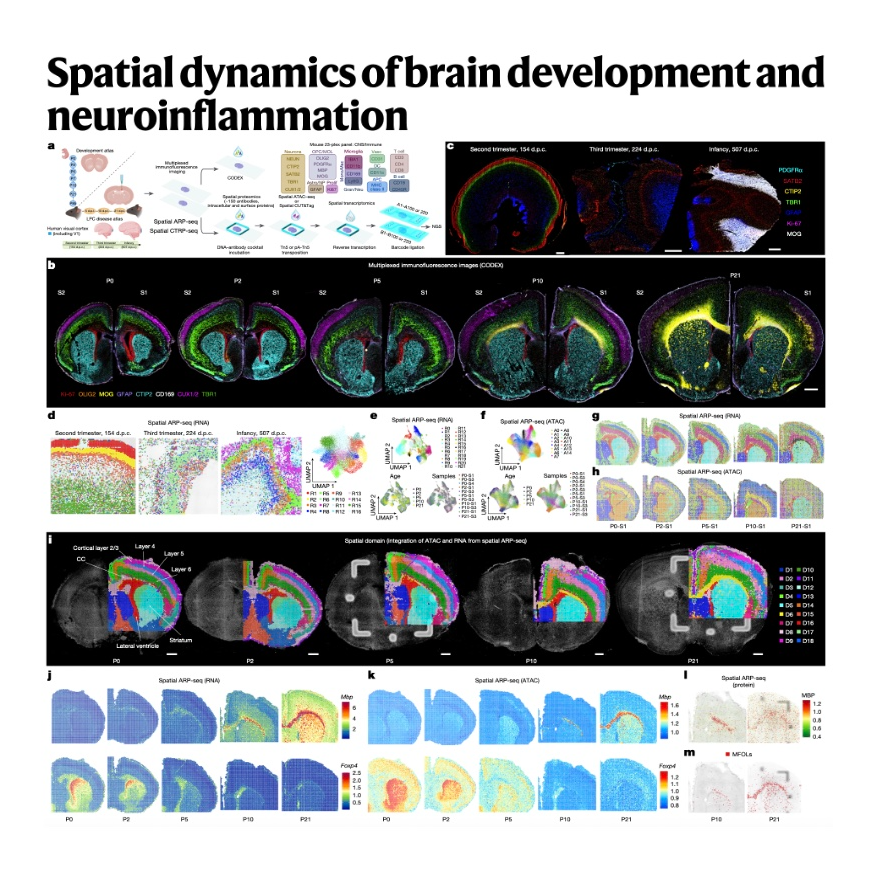
This research introduces DBiT-based spatial tri-omic technologies, specifically spatial ARP-seq and spatial CTRP-seq, to simultaneously map the epigenome, transcriptome, and proteome within the same tissue section. Researchers applied these tools to study mouse and human brain development, revealing how chromatin priming and gene expression coordinate the complex layering of the cerebral cortex. The study highlights a lateral-to-medial progression of myelination in the corpus callosum, steered by specific projection neuron tracts. Furthermore, the authors investigated neuroinflammation using a demyelination model, identifying distinct immune cell states and a delayed inflammatory response in distal white matter. By integrating multiple molecular layers, the sources provide a high-resolution atlas of the mechanisms governing central nervous system maturation and its reaction to injury.References: Zhang D, Rubio Rodríguez-Kirby L A, Lin Y, et al. Spatial dynamics of brain development and neuroinflammation[J]. Nature, 2025, 647(8088): 213-227.
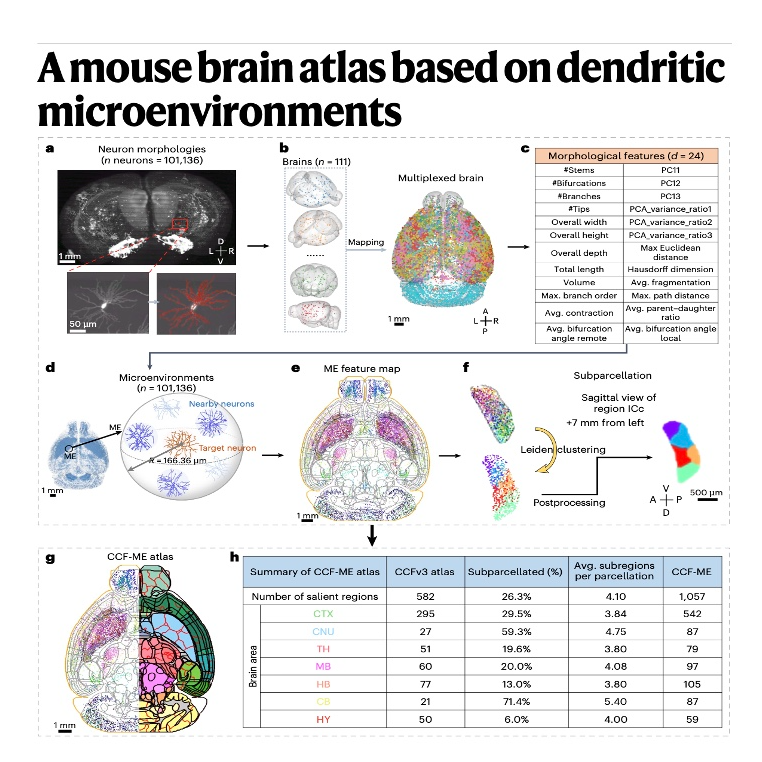
Researchers have developed a new mouse brain atlas, termed CCF-ME, by focusing on the dendritic microenvironments of over 100,000 neurons. Unlike traditional mapping methods that rely on cell density or gene expression, this approach categorizes brain regions based on the local structural patterns and branching shapes of individual nerve cells. This method revealed a significantly more detailed anatomical organization, nearly doubling the number of identifiable subregions compared to previous standards like the Allen Common Coordinate Framework. The study discovered that these local dendritic arrangements are strong predictors of long-range connectivity, showing that neurons with similar microenvironments often link to the same distant targets. Furthermore, the atlas provides a high-resolution complement to spatial transcriptomics, offering a clearer understanding of how fine-grained brain structure relates to biological function.References: Liu Y, Zhao S, Yun Z, et al. A mouse brain atlas based on dendritic microenvironments[J]. Nature Neuroscience, 2025: 1-12.
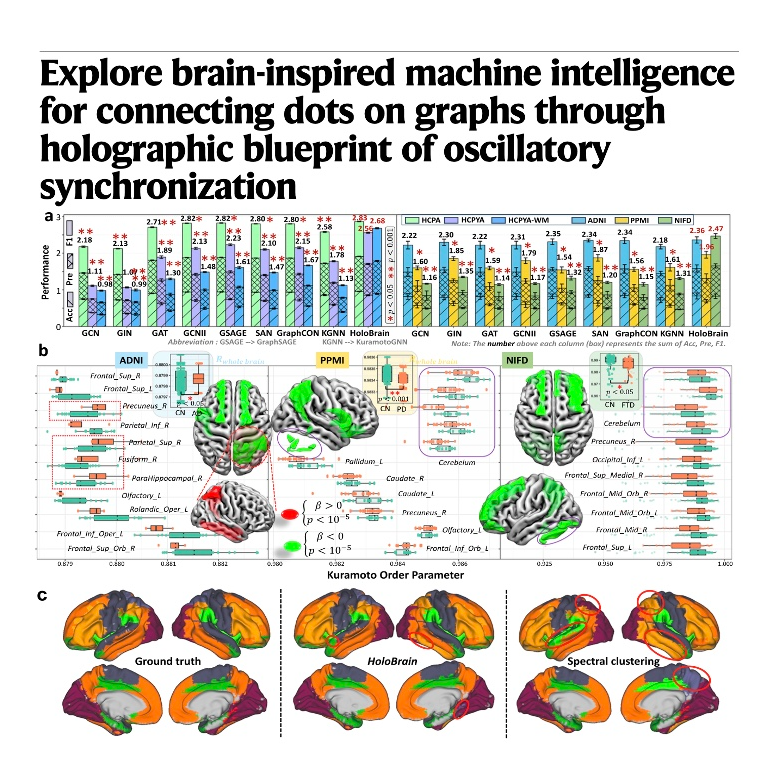
This research introduces HoloGraph, a novel framework that uses the Kuramoto model of coupled oscillators to improve how machines learn from complex data networks. By treating individual nodes as rhythmic oscillators that synchronize over time, the system mimics the way the human brain integrates diverse sensory information through neural oscillations. The study demonstrates that this method outperforms existing models in task recognition and remains stable even in very deep networks where other systems typically fail. Beyond computer science, the authors apply this technology to neuroscience, identifying how synchronization patterns change in patients with Alzheimer’s and Parkinson’s diseases. Ultimately, the work bridges the gap between dynamic biological systems and artificial intelligence to provide a more sophisticated tool for analyzing healthy and pathological brain aging.References: Dan T, Ding J, Wu G. Explore brain-inspired machine intelligence for connecting dots on graphs through holographic blueprint of oscillatory synchronization[J]. Nature Communications, 2025, 16(1): 9425.
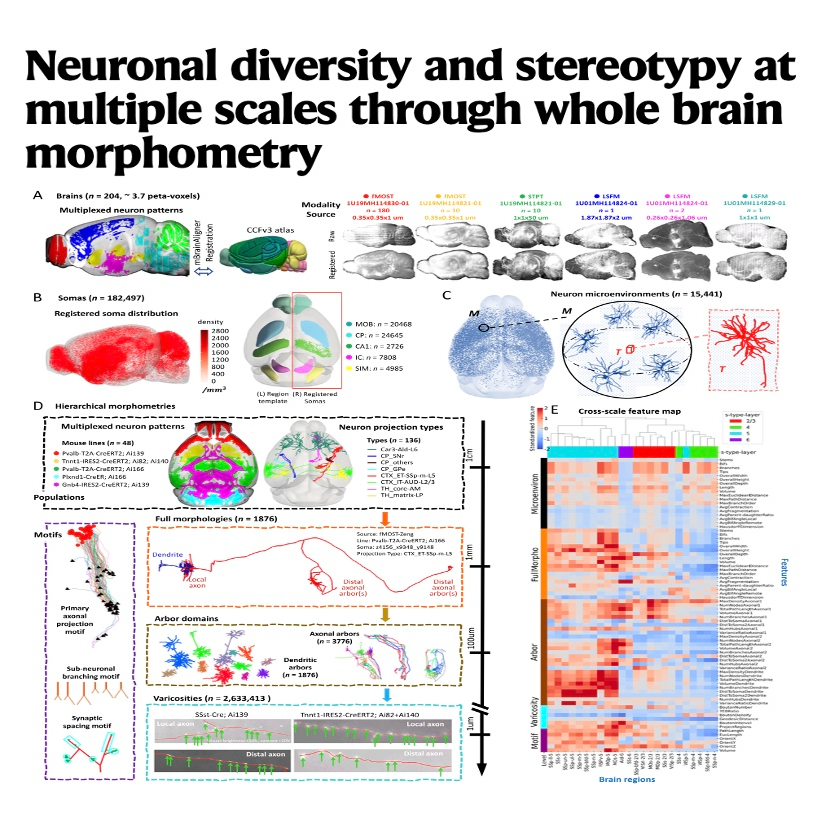
This study presents a comprehensive multi-scale morphometry of the mouse brain, analyzing neuronal structures from individual synapses to whole-organ circuits. Researchers developed the Collaborative Augmented Reconstruction (CAR) platform to map over 180,000 somas and nearly 2,000 full neuronal morphologies. By quantifying diversity and stereotypy, the authors identified conserved projection motifs and dendritic microenvironments that align with established anatomical boundaries. Their work reveals that while global axonal tracts exhibit high diversity, local structures like varicosities follow highly predictable patterns. This open-source framework provides the neuroscience community with quantitative descriptors to better understand the relationship between neuronal shape and brain function. Ultimately, the resource bridges the gap between single-neuron data and the macroscopic organization of the mammalian nervous system.References: Liu Y, Jiang S, Li Y, et al. Neuronal diversity and stereotypy at multiple scales through whole brain morphometry[J]. Nature communications, 2024, 15(1): 10269.
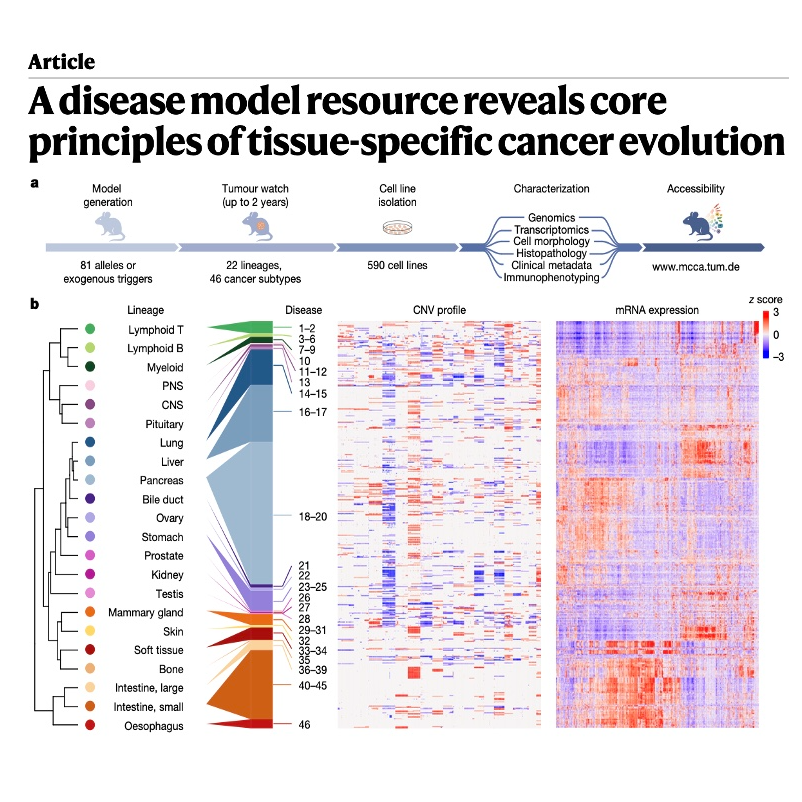
This research introduces the Mouse Cancer Culture Collection (MCCA), a massive repository of nearly 600 cell lines representing 46 distinct cancer types. By utilizing this resource, scientists identified that cancer evolution is a deterministic process shaped heavily by the specific tissue in which it originates. A key focus is the KRAS oncogene, where findings show that different organs require specific dosage levels of the mutation to trigger tumor growth or cellular reprogramming. The study further demonstrates that the CDKN2A tumor suppressor behaves differently across tissues due to varying epigenetic states, which explains why some organs are more resistant to cancer than others. Ultimately, these results provide a comprehensive framework for understanding the genomic trajectories of human disease through highly accurate mouse models.References: Mueller S, de Andrade Krätzig N, Tschurtschenthaler M, et al. A disease model resource reveals core principles of tissue-specific cancer evolution[J]. Nature, 2026: 1-12.
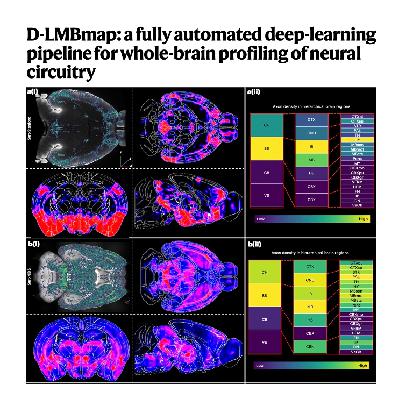
The paper introduces D-LMBmap, an integrated open-source software suite designed for the automated 3D mapping of whole-brain neuronal connectivity using light-sheet fluorescence microscopy. This framework addresses critical bottlenecks in neuroimaging by offering advanced tools for automated axon segmentation, brain-style transfer, and multiscale 3D registration. By utilizing deep neural networks and innovative data augmentation, the system significantly reduces the manual labor typically required for annotating complex axonal projections and identifying anatomical regions. It also employs a multi-constraint registration strategy that ensures high accuracy when aligning experimental samples with standard brain atlases, even in damaged tissues or across different imaging modalities. Ultimately, this comprehensive pipeline enables high-throughput, mesoscale profiling of diverse neuron types, providing researchers with a more efficient method for quantifying and visualizing intricate brain-wide circuits.References: Li Z, Shang Z, Liu J, et al. D-LMBmap: a fully automated deep-learning pipeline for whole-brain profiling of neural circuitry[J]. Nature Methods, 2023, 20(10): 1593-1604.
This paper examine the dural sinuses as a specialized interface that manages both fluid dynamics and immune surveillance in the brain. Researchers identified sinus endothelial cells (SECs) as highly active regulators that adjust their boundaries to let immune cells move between the blood and the surrounding perisinus space. This process, often appearing as cellular ruffling, is essential for defending the central nervous system against viral infections and other circulating pathogens. The study highlights that RAMP2 signaling and specific adhesion molecules are critical for maintaining this protective barrier's flexibility. By comparing mouse models with human imaging, the authors suggest these neuroimmune mechanisms are largely conserved across species. Ultimately, understanding how these vessels balance blood flow and pathogen defense may lead to new treatments for various neurological diseases.References: Monaghan K L, Zanluqui N G, Su Y, et al. Highly dynamic dural sinuses support meningeal immunity[J]. Nature, 2026: 1-10.
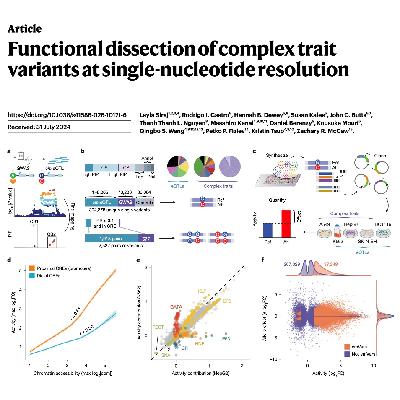
This study uses high-throughput reporter assays to bridge the gap between genetic associations and the actual biological mechanisms of disease. Researchers employed massively parallel reporter assays (MPRA) to screen over 300,000 DNA sequences, identifying specific causal variants that regulate gene expression across various human tissues. Through saturation mutagenesis, they mapped how single-nucleotide changes disrupt or create transcription factor binding sites, revealing both simple and complex regulatory patterns. The findings include evidence of regulatory epistasis, where multiple genetic variations interact non-additively to influence traits like blood pressure and cholesterol. By combining these experimental maps with predictive neural networks, the authors provide a comprehensive framework for understanding how non-coding mutations contribute to human health. This research ultimately transforms statistical correlations from large biobanks into functional insights at single-nucleotide resolution.References: Siraj L, Castro R I, Dewey H B, et al. Functional dissection of complex trait variants at single-nucleotide resolution[J]. Nature, 2026: 1-11.
This study investigates how brain connectivity patterns can identify individuals at ultra-high risk (UHR) for psychosis and predict their everyday social and occupational functioning. Researchers utilized resting-state fMRI and machine learning to demonstrate that both increased and decreased connectivity—particularly involving the thalamus as a central hub—accurately distinguish UHR individuals from healthy peers. Notably, the same neural networks that signal a risk for psychosis also correlate with a person’s level of functioning, regardless of whether they eventually develop a full psychotic disorder. The findings suggest that hyper-connectivity in specific circuits may reflect a compensatory effort to maintain performance despite underlying neurological strain. Ultimately, the research establishes functional connectivity as a significant biomarker for the clinical impairments and biological realities associated with the early stages of mental illness.References: Ambrosen K S, Kristensen T D, Glenthøj L B, et al. Whole-brain functional connectivity predicts ultra-high risk for psychosis status and level of functioning[J]. Schizophrenia, 2026.
This research investigates how the human brain's hierarchical structure influences its response to electrical stimulation by combining invasive and non-invasive electrophysiology with advanced mathematical modeling. By analyzing data from 36 patients, the study identifies a spatial gradient of excitability, where high-order cognitive networks exhibit much stronger and more integrated responses than low-order sensory regions. Using "virtual dissections" in a computational model, the authors demonstrate that these heightened responses in high-order networks are causally dependent on recurrent feedback loops from the rest of the brain. In contrast, simpler sensory networks operate with greater functional segregation, relying primarily on internal activity rather than global communication. These findings reveal that a region’s position within the cortical hierarchy dictates its information-processing strategy and its level of network integration. Ultimately, this work provides a mechanistic framework for understanding brain dynamics and could lead to more effective, personalized neuromodulation therapies.References: Momi D, Wang Z, Parmigiani S, et al. Stimulation mapping and whole-brain modeling reveal gradients of excitability and recurrence in cortical networks[J]. Nature Communications, 2025, 16(1): 3222.