Discover Cellular and Molecular Biology for Research
Cellular and Molecular Biology for Research

Cellular and Molecular Biology for Research
Author: Ahmadreza Gharaeian
Subscribed: 49Played: 271Subscribe
Share
© Ahmadreza Gharaeian
Description
Cellular and Molecular Biology for Research is the podcast where complex textbooks stop gathering dust and start making sense. Each episode breaks down the dense chapters of cellular and molecular biology—DNA, signaling pathways, protein folding, experimental techniques—into clear explanations for students, early-career researchers, or anyone who wants to actually understand the science instead of just memorizing it. Think of it as your study buddy who reads the heavy stuff, translates the jargon, and hands you the key concepts (with a little less pain and a lot more clarity).
54 Episodes
Reverse
Your skin, muscles, and joints are constantly talking — and your brain is always listening. In this episode, we dive into the somatic sensory system, the network that lets you feel a soft breeze, a burning flame, or the sharp sting of a pinprick.Unlike sight or hearing, this system isn’t confined to one organ — it’s everywhere. It’s how you sense touch, temperature, pain, and body position, working together to map your body’s reality in real time. From the pleasure of warmth to the lifesaving agony of pain, these sensations define what it means to live in a body.Join us as we uncover how your nerves translate texture, pressure, and even itch into the rich, continuous language of feeling — a story told entirely through electricity and experience.
In this episode, we dive into the twin marvels of the auditory and vestibular systems — the senses that let us hear the world and stay upright in it. From the crash of a wave to the whisper of a friend, your brain turns invisible vibrations into vivid perception. Meanwhile, your inner ear quietly works overtime, keeping you balanced and your vision steady even as your head moves.We’ll break down how sound waves and head motion are transformed into neural code, how your brainstem and thalamus orchestrate this sensory duet, and why hearing and equilibrium—though seemingly worlds apart—share deep evolutionary roots.
Vision is our window to both the microscopic and the cosmic — from spotting a mosquito on your nose to glimpsing galaxies millions of light-years away. Yet for all its apparent simplicity, seeing is one of the most complex feats biology has ever pulled off.In this episode, we peel back the layers of how the brain turns light — mere electromagnetic waves bouncing through space — into meaning. You’ll discover how evolution built the eye as a living camera, and how the retina, a literal piece of the brain tucked inside your eyeball, begins processing images before they ever reach your cortex.We’ll explore how the optic nerves ferry signals to brain regions that set your internal clock, move your eyes, and ultimately let you perceive the world in color, contrast, and motion. From the low-light world of night vision to the dazzling spectrum of daylight, every photon that hits your retina becomes part of the grand neural symphony of sight.And as it turns out, vision didn’t just help us survive — it helped us imagine, predict, create, and paint our understanding of reality itself.
Long before brains existed, life was already listening — not to sounds or sights, but to chemicals. From single-celled bacteria to humans, survival has always depended on detecting the molecules that mean food, danger, or love. In this episode, we dive into the most ancient and universal senses of all: taste and smell.We’ll explore how evolution shaped our ability to sense the world through chemistry — from bacteria swimming toward nutrients to humans savoring the sweetness of honey or catching the scent of pizza. You’ll learn how chemoreceptors scattered throughout the body detect everything from the flavor of food to the acidity in our muscles, and how our gustatory and olfactory systems work together to create the experience we call flavor.But taste and smell do more than please the palate — they’re deeply tied to emotion, memory, hunger, and even desire. These senses connect the oldest parts of our brain to the most primal parts of our behavior.Join us as we uncover how the chemical world outside becomes meaning inside — and how every breath, bite, and scent speaks the universal language of life itself.
Before we dive into how the brain works, we need to know how it’s built. In this episode, we open the Illustrated Guide to the Brain — your map to the physical landscape of the nervous system.We’ll explore the brain not just as a concept, but as a real, three-dimensional structure with surfaces, sections, and systems that all fit together inside the skull. From the folds of the cerebral cortex to the deep cores of the brainstem and spinal cord, this guided tour will show how anatomy lays the groundwork for everything the nervous system does.You’ll learn how neuroscientists divide the brain into functional systems — like the visual, olfactory, and auditory networks — and how these systems connect into one coordinated whole. We’ll also touch on the cranial nerves, the autonomic nervous system, and the blood vessels that keep the brain alive and working.Think of this as a traveler’s guide to the brain’s terrain — a way to learn the names and landmarks before we start exploring their functions in depth.
Your thoughts, movements, and moods all depend on chemistry — specifically, the brain’s breathtakingly precise neurotransmitter systems. In this episode, we dive into the molecules that make neurons talk, and the elegant machinery that keeps those conversations going.We’ll revisit the pioneers of neurochemistry, from Otto Loewi, who discovered acetylcholine and proved that neurons communicate with chemicals, to Henry Dale, who gave us the language we still use today — cholinergic, noradrenergic, glutamatergic, GABAergic. Each neurotransmitter system isn’t just a single molecule; it’s an entire operation: the enzymes that make it, the vesicles that store it, the transporters that recycle it, and the receptors that respond to it.From amino acids to amines to peptides, these tiny messengers define how the brain controls everything from muscle contraction to mood regulation. Understanding them is key to unlocking how drugs, disorders, and even our own emotions shape neural activity.Join us as we explore the variety, precision, and beauty of the brain’s chemical code — the systems that turn electricity into emotion, thought into action, and chemistry into consciousness
Your thoughts, movements, and moods all depend on chemistry — specifically, the brain’s breathtakingly precise neurotransmitter systems. In this episode, we dive into the molecules that make neurons talk, and the elegant machinery that keeps those conversations going.We’ll revisit the pioneers of neurochemistry, from Otto Loewi, who discovered acetylcholine and proved that neurons communicate with chemicals, to Henry Dale, who gave us the language we still use today — cholinergic, noradrenergic, glutamatergic, GABAergic. Each neurotransmitter system isn’t just a single molecule; it’s an entire operation: the enzymes that make it, the vesicles that store it, the transporters that recycle it, and the receptors that respond to it.From amino acids to amines to peptides, these tiny messengers define how the brain controls everything from muscle contraction to mood regulation. Understanding them is key to unlocking how drugs, disorders, and even our own emotions shape neural activity.Join us as we explore the variety, precision, and beauty of the brain’s chemical code — the systems that turn electricity into emotion, thought into action, and chemistry into consciousness.
We’ve seen how a thumbtack to the foot can trigger an electrical storm in your nerves — but how does that signal jump from one neuron to the next? Welcome to the synapse, the tiny but mighty junction where information changes hands.In this episode, we trace the story from the late 1800s, when scientists first realized neurons don’t just touch — they communicate. Early researchers like Charles Sherrington gave this mysterious meeting point a name, while others debated whether neurons talked through electricity or chemistry.We’ll follow the experiments that settled the score — from Otto Loewi’s famous frog heart experiment that revealed chemical messengers, to Bernard Katz’s work showing how nerve impulses trigger neurotransmitter release, and John Eccles’ discovery that most brain synapses rely on chemical signaling.Today, we know that synaptic transmission is at the heart of everything the nervous system does — from reflexes to memory, emotions to mental illness.Join us as we unpack how these tiny connections create the grand symphony of the brain: how neurotransmitters are made, stored, and released, and how every signal you think, feel, or remember begins at the space between two neurons.
Your brain speaks in electricity — tiny, rapid bursts called action potentials. In this episode, we break down the signal that carries information through your nervous system at lightning speed. Normally, a neuron’s interior is slightly negative compared to the outside — but when an action potential hits, that balance flips in a split second, and the inside becomes positive.This brief electrical surge, also known as a spike or nerve impulse, races along the axon without losing strength. Every thought, movement, and sensation you have depends on the frequency and pattern of these impulses — the brain’s own version of Morse code.Join us as we explore how neurons generate and send these powerful signals, and how a single pulse of electricity becomes the foundation for everything your nervous system does
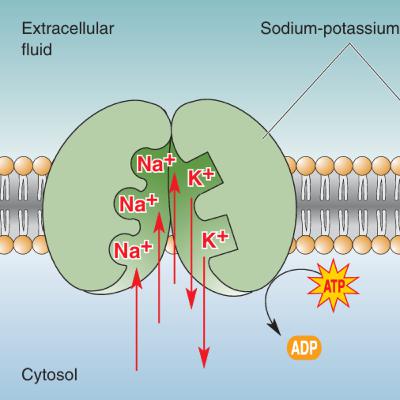
Ever wonder what’s happening inside your body when you step on a thumbtack and instantly yank your foot away? In this episode, we dive into the electrifying world of your nervous system — literally. From the first spark of pain at your skin to the lightning-fast signals racing up your spinal cord, we unpack how neurons collect, process, and transmit information.You’ll learn how the brain’s communication lines — neurons — send signals not through copper wires, but through charged atoms called ions, and how they’ve evolved a clever trick called the action potential to keep those signals strong and fast. We’ll also uncover the secret “battery” that powers every thought and movement: the resting membrane potential.Join us as we explore how a tiny voltage difference across a cell’s membrane builds the foundation for everything your brain and body can do — from reflexes to reasoning.
The historical foundations of neuroscience were laid by numerous individuals over many generations. Today, researchers at various levels of analysis and employing diverse technologies are making significant strides in uncovering the brain's functions. The results of these endeavors form the basis of this textbook. The primary aim of neuroscience is to comprehend how nervous systems operate. Valuable insights can often be gained from observing the brain’s activity indirectly. Since behavior reflects brain activity, careful behavioral measurements provide information about the brain's functional capabilities and limitations. Computational models that replicate the brain’s computational properties allow us to explore how such properties emerge. By recording brain waves from the scalp, we can investigate the electrical activity of different brain regions during various behavioral states. Advanced imaging techniques now enable researchers to examine the structure of the living brain in situ, while even more sophisticated methods reveal which brain areas become active under specific conditions. However, despite the advancements in noninvasive methods, these approaches cannot entirely replace direct experimentation with living brain tissue. To interpret remote signals accurately, it is essential to understand how they are generated and their significance. A comprehensive understanding of brain function requires examining its contents—neuroanatomically, neurophysiologically, and neurochemically. The current pace of neuroscience research is remarkable, fueling for new treatments for the many debilitating nervous system disorders affecting millions annually. Yet, despite centuries of progress, including recent decades of advancement, a complete understanding of the brain’s extraordinary abilities remains a distant goal. Nevertheless, this ongoing journey continues to inspire hope and discovery.
Functional genomics focuses on analyzing the expression of numerous genes. One branch of this field is transcriptomics, which examines transcriptomes—all the RNA transcripts produced by an organism at a specific time. A common approach in transcriptomics involves the creation of DNA microarrays or microchips containing thousands of cDNAs or oligonucleotides. These arrays are hybridized with labeled RNAs (or their corresponding cDNAs) from cells, and the hybridization intensity at each spot indicates the expression level of the corresponding gene. This method enables the simultaneous analysis of the timing and location of expression for multiple genes.Serial Analysis of Gene Expression (SAGE) identifies which genes are expressed in a particular tissue and measures their expression levels. It works by generating short gene-specific tags from cDNAs, ligating them between linkers, and sequencing the ligated tags to determine gene expression and abundance. Cap Analysis of Gene Expression (CAGE) provides similar data but focuses on the 5'-ends of mRNAs, enabling the identification of transcription start sites and aiding in the localization of promoters.High-density transcriptional mapping of entire chromosomes has revealed that most sequences in cytoplasmic polyadenylated RNAs originate from non-exon regions of ten human chromosomes. Additionally, nearly half of the transcription from these chromosomes is nonpolyadenylated. These findings suggest that the majority of stable nuclear and cytoplasmic transcripts derive from regions outside exons, which may explain significant differences between species, such as humans and chimpanzees, whose exons are nearly identical.
Several approaches are available for identifying genes within a large, unsequenced DNA region. One method is the exon trap, which employs a specialized vector to selectively clone exons. Another involves using methylation-sensitive restriction enzymes to locate CpG islands—DNA regions containing unmethylated CpG sequences. Prior to the genomics era, geneticists mapped the Huntington disease gene (HD) to a region near the end of chromosome 4, subsequently using an exon trap to identify the gene itself.Advancements in automated DNA sequencing methods have enabled molecular biologists to determine the base sequences of various organisms, from simple phages and bacteria to yeast, plants, animals, and humans. In the Human Genome Project, much of the mapping work utilized yeast artificial chromosomes (YACs), which are vectors containing a yeast origin of replication, a centromere, and two telomeres. These vectors can accommodate foreign DNA up to 1 million base pairs long, which replicates alongside the YAC. However, due to their superior stability and ease of use, bacterial artificial chromosomes (BACs) became the preferred tool for sequencing. BACs, derived from the F plasmid of E. coli, can accept DNA inserts up to approximately 300 kilobases, with an average insert size of about 150 kilobases.Mapping large genomes, such as the human genome, requires a set of landmarks (markers) to determine the positions of genes. While genes themselves can serve as markers, most markers consist of anonymous DNA segments like RFLPs, VNTRs, STSs (including ESTs), and microsatellites. Restriction fragment length polymorphisms (RFLPs) are variations in the lengths of DNA fragments produced by cutting DNA from different individuals with a restriction enzyme, often caused by the presence or absence of specific restriction sites.
Transposable elements, also known as transposons, are DNA segments capable of moving from one location to another within the genome. Some transposable elements replicate during the process, leaving one copy in the original position and inserting a new copy at a different site, while others move without replication, vacating the original site entirely. Bacterial transposons can be categorized as follows: (1) insertion sequences, such as IS1, which consist solely of the genes required for transposition and are flanked by inverted terminal repeats; and (2) transposons like Tn3, which resemble insertion sequences but include at least one additional gene, often conferring antibiotic resistance.Eukaryotic transposons exhibit diverse replication strategies. DNA transposons, such as Ds and Ac in maize or the P elements in Drosophila, function similarly to bacterial DNA transposons like Tn3.The immunoglobulin genes in mammals undergo rearrangement through a mechanism analogous to transposition. Vertebrate immune systems generate immense diversity in immunoglobulin production by assembling genes from two or three components selected from a heterogeneous pool. This process, called V(D)J recombination, relies on recombination signal sequences (RSSs) that include a heptamer and a nonamer separated by either 12-bp or 23-bp spacers. Recombination occurs exclusively between a 12 signal and a 23 signal, ensuring the incorporation of only one of each type of coding region into the assembled gene. Key players in human V(D)J recombination are RAG1 and RAG2, which create single-strand nicks in DNA adjacent to a 12 or 23 signal. This triggers a transesterification reaction where the newly formed 3'-hydroxyl group attacks the opposite strand, leading to a break and forming a hairpin at the end of the coding segment.
Homologous recombination is vital for life. In eukaryotic meiosis, it ensures proper separation of homologous chromosomes by locking them together and promotes genetic diversity in offspring by scrambling parental genes. In all life forms, it plays a crucial role in managing DNA damage. In E. coli, homologous recombination via the RecBCD pathway starts with the invasion of duplex DNA by single-stranded DNA from another duplex that has undergone a double-stranded break. This process begins with RecBCD's nuclease and helicase activities, which generate a free end by preferentially nicking DNA at Chi sites. The invading strand is then coated with RecA and SSB. RecA facilitates the pairing of the invading strand with its complementary homologous DNA, forming a D-loop, while SSB enhances recombination by melting secondary structures and preventing RecA from trapping such structures, which could inhibit subsequent strand exchange. Following this, RecBCD likely nicks the D-loop strand, creating a branched intermediate known as a Holliday junction. The RuvA–RuvB helicase catalyzes branch migration, moving the crossover of the Holliday junction to a favorable resolution site. Finally, RuvC resolves the Holliday junction by nicking two of its strands, producing either noncrossover recombinants with heteroduplex patches or two crossover recombinant DNAs.Meiotic recombination in yeast begins with double-stranded breaks (DSBs) created by two Spo11 molecules. These molecules work together to cleave both DNA strands at closely spaced sites through transesterification reactions involving active site tyrosines. This reaction forms covalent bonds between Spo11 and the newly created DSBs. Spo11 is subsequently released.
Primer synthesis in E. coli involves the primosome, which consists of the DNA helicase DnaB and the primase DnaG. The assembly of the primosome at the origin of replication, oriC, proceeds as follows: DnaA binds to oriC at specific sites known as dnaA boxes and collaborates with RNA polymerase and HU protein to melt a DNA region adjacent to the leftmost dnaA box. Subsequently, DnaB associates with the open complex and promotes the binding of the primase to complete the primosome. The primosome remains attached to the replisome, repeatedly initiating Okazaki fragment synthesis on the lagging strand. Additionally, DnaB exhibits helicase activity, unwinding the DNA as the replisome advances.In the case of the SV40 origin of replication, it is located adjacent to the viral transcription control region. Replication initiation relies on the viral large T antigen, which binds within the 64-bp minimal ori at two adjacent sites. This antigen also possesses helicase activity, creating a replication bubble within the minimal ori. Priming is performed by a primase associated with the host DNA polymerase α.Yeast origins of replication are found within autonomously replicating sequences (ARSs), which consist of four key regions: A, B1, B2, and B3. Region A, a 15-bp sequence, contains an 11-bp consensus sequence that is highly conserved across ARSs. Region B3 may contribute to a critical DNA bend within ARS1.The pol III holoenzyme synthesizes DNA at a rate of approximately 730 nucleotides per second in vitro, slightly slower than the nearly 1000 nucleotides per second observed in vivo. This enzyme is highly processive both in vitro and in vivo. The pol III core (αε or αεθ) alone lacks processivity and can only replicate short DNA segments before dissociating from the template. However, when combined with the β-subunit, the core achieves processive replication at a rate approaching 1000 nucleotides per second. The β-subunit forms a dimer that takes on a ring-like structure, encircling the DNA.
Several principles govern DNA replication across most organisms: (1) Double-stranded DNA replicates in a semiconservative manner, where the parental strands separate and serve as templates for the synthesis of new, complementary strands. (2) DNA replication in E. coli and other organisms is at least semidiscontinuous. One strand, often considered to replicate continuously in the direction of the replication fork's movement, may actually replicate discontinuously. The other strand replicates discontinuously, forming 1–2 kb Okazaki fragments in the opposite direction, allowing both strands to be synthesized in the 5'→3' direction. (3) DNA replication initiation requires a primer. In E. coli, Okazaki fragments are initiated with RNA primers that are 10–12 nucleotides long. (4) Most bacterial and eukaryotic DNAs replicate bidirectionally, though some, like ColE1, replicate unidirectionally.Circular DNAs can replicate via the rolling circle mechanism, where one strand of the double-stranded DNA is nicked, and the 3'-end is extended using the intact strand as a template. This process displaces the 5'-end, and in phage λ, the displaced strand serves as a template for discontinuous, lagging strand synthesis.Pol I is a highly versatile enzyme with three distinct activities: DNA polymerase, 3'→5' exonuclease, and 5'→3' exonuclease. The first two activities reside on a large domain of the enzyme, while the third is on a smaller, separate domain. The large domain, known as the Klenow fragment, can be isolated through mild protease treatment, yielding two protein fragments with all three activities intact. The structure of the Klenow fragment includes a wide cleft for DNA binding, with the polymerase active site located far from the 3'→5' exonuclease active site.Among the three DNA polymerases in E. coli—Pol I, Pol II, and Pol III—only Pol III is essential for replication.
X-ray crystallography studies on bacterial ribosomes with and without tRNAs have revealed that tRNAs occupy the cleft between the two subunits. They interact with the 30S subunit through their anticodon ends and with the 50S subunit through their acceptor stems. The binding sites for tRNAs primarily consist of rRNA. The anticodons of tRNAs in the A and P sites come into close proximity, allowing base-pairing with adjacent codons in the mRNA bound to the 30S subunit, as the mRNA bends 45 degrees between the two codons. The acceptor stems of tRNAs in the A and P sites also approach each other closely—within just 5 Å—within the peptidyl transferase pocket of the 50S subunit, where twelve contacts between ribosomal subunits are visible.The crystal structure of the E. coli ribosome reveals two conformations that differ due to rigid body motions of ribosomal domains relative to each other. Specifically, the head of the 30S particle rotates by 6 degrees and by 12 degrees when compared to the T. thermophilus ribosome. This rotation is likely part of the ratchet-like motion of the ribosome during translocation.The E. coli 30S subunit comprises a 16S rRNA and 21 proteins (S1–S21), while the 50S subunit contains a 5S rRNA, a 23S rRNA, and 34 proteins (L1–L34). Eukaryotic cytoplasmic ribosomes are larger and include more RNAs and proteins than their prokaryotic counterparts. Sequence studies of 16S rRNA proposed its secondary structure (intramolecular base pairing), which has been confirmed by X-ray crystallography studies. These studies reveal a 30S subunit with extensively base-paired 16S rRNA, whose shape essentially defines the particle's overall structure. Additionally, X-ray crystallography studies have identified the locations of most 30S ribosomal proteins.The 30S ribosomal subunit serves two primary roles. It facilitates accurate decoding of mRNA and contributes to the overall function of the ribosome during translation.
Messenger RNAs are read in the 5' to 3' direction, which is the same direction in which are synthesized. Proteins are synthesized from the amino terminus to the carboxyl terminus, meaning the amino-terminal amino acid is added first. The genetic code consists of three-base sequences called codons in mRNA, which instruct the ribosome to incorporate specific amino acids into a polypeptide. The code nonoverlapping, meaning each base is part of only one codon, and it lacks gaps or commas, with every base in the coding region of an mRNA being part of a codon. There are 64 codons in total, three of which are stop signals, while the remaining codons encode amino acids, making the code highly degenerate. The degeneracy of the genetic code is partially managed by isoaccepting tRNA species that bind the same amino acid but recognize different codons. Additionally, wobble pairing allows the third base of a codon to deviate slightly from its normal position, forming non-Watson–Crick base pairs with the anticodon. This enables a single aminoacyl-tRNA to pair with multiple codons. Wobble pairs include G–U (or I–U) and I–A. The genetic code is not strictly universal. In certain eukaryotic nuclei, mitochondria, and at least one bacterium, codons that serve as termination signals in the standard genetic code can instead encode amino acids such as tryptophan and glutamine. In some mitochondrial genomes, the meaning of codons is altered, switching from one amino acid to another. Despite these deviations, the altered codes remain closely related to the standard genetic code from which they likely evolved. Elongation occurs in three steps: (1) EF-Tu, bound with GTP, delivers an aminoacyl-tRNA to the ribosomal A site. (2) Peptidyl transferase forms a peptide bond between the peptide in the P site and the newly arrived aminoacyl-tRNA in the A site, extending the peptide by one amino acid and shifting it to the A site. (3) EF-G, in conjunction with GTP, translocates the growing peptide.
Two critical events precede protein synthesis. First, aminoacyl-tRNA synthetases attach amino acids to their respective tRNAs with high specificity through a two-step reaction that begins with the activation of the amino acid using AMP, derived from ATP. Second, ribosomes must dissociate into their subunits at the conclusion of each translation cycle. In bacteria, this dissociation is actively facilitated by RRF and EF-G, while IF3 binds to the free 30S subunit, preventing its reassociation with the 50S subunit to form a complete ribosome.The initiation codon in prokaryotes is typically AUG but can also be GUG or, more rarely, UUG. The initiating aminoacyl-tRNA is N-formyl-methionyl-tRNAfMet. N-formyl-methionine (fMet) is the first amino acid incorporated into a polypeptide chain, although it is often removed during protein maturation. The 30S initiation complex is formed by the association of a free 30S ribosomal subunit with mRNA and fMet-tRNAfMet. This binding depends on base pairing between the Shine-Dalgarno sequence, located just upstream of the initiation codon in mRNA, and a complementary sequence at the 3'-end of the 16S rRNA. IF3 mediates this interaction with the assistance of IF1 and IF2, which are all bound to the 30S subunit at this stage.IF2 plays a central role in promoting the binding of fMet-tRNAfMet to the 30S initiation complex, while the other two initiation factors provide essential support. GTP is required for IF2 binding under physiological IF2 concentrations, though it is not hydrolyzed during this process. The complete 30S initiation complex consists of one 30S ribosomal subunit, one molecule each of mRNA, fMet-tRNAfMet, GTP, IF1, IF2, and IF3. GTP hydrolysis occurs after the 50S subunit joins the 30S complex to form the functional 70S initiation complex.