Discover Uncapped: Stories from the Grey Market
Uncapped: Stories from the Grey Market

Uncapped: Stories from the Grey Market
Author: krysia
Subscribed: 0Played: 0Subscribe
Share
© krysia
Description
Uncapped, the podcast that dives deep into the underground world of grey market peptides, research compounds, and the Discord and Telegram communities that operate just outside the lines.
Each episode blends real events, from chaotic group buys to vendor takedowns, with educational breakdowns, industry interviews, and sharp commentary from the people who live in this unregulated world. Some stories are dramatic. Some are absurd. All are true, or at least, true enough to make you rethink what’s really going on behind the scenes of your favourite “trusted vendor.”
Each episode blends real events, from chaotic group buys to vendor takedowns, with educational breakdowns, industry interviews, and sharp commentary from the people who live in this unregulated world. Some stories are dramatic. Some are absurd. All are true, or at least, true enough to make you rethink what’s really going on behind the scenes of your favourite “trusted vendor.”
60 Episodes
Reverse
Scams, impersonators, vendor disputes, disappearing chats, and a collaboration offer I definitely wasn’t expecting.This episode is a real-time look at what one day in the grey zone actually looks like for me, narrated in my own voice.
What really happens when tirzepatide and vitamin B12 are mixed in the same vial?In this deep dive, we break down the science behind Eli Lilly’s recent warning and preprint, which identified a previously unknown tirzepatide–B12 molecular complex in compounded samples, in some cases making up a significant portion of the vial. The analysis also revealed major potency variation, with some products testing far below their labelled strength.But the real question isn’t just what was found, it’s what it actually means.We explore:how and why molecules interact differently in a shared vial vs the human bodywhat Lilly’s analytical data proves, and what it doesn’twhy formulation conditions (pH, concentration, solvent) can drive structural changeand how theoretical risks like aggregation and immunogenicity should be interpretedThis episode separates chemical possibility from clinical reality, and explains why understanding where interactions occur is key to understanding whether they matter at all.
What their data shows and what it still doesn't explain.What happens when a blockbuster drug meets a common vitamin?In this deep dive, we unpack Eli Lilly’s warning about tirzepatide mixed with vitamin B12, and the science behind the controversy.A newly released preprint shows that when the two are combined, they can form a previously unknown molecular complex, detected in real world samples. Some products were also found to be significantly under strength, raising broader concerns about quality in the secondary market.But while the chemistry is real, the key question remains unanswered:does any of this actually matter for patients?We break down:what Lilly’s data actually proveshow the molecules interact at a structural levelwhy lab findings don’t automatically translate to real-world riskand how this fits into the wider $100B GLP-1 market battleThis isn’t just a story about one drug or one impurity, it’s about the gap between analytical evidence and clinical reality, and what happens when patients are caught in the middle.
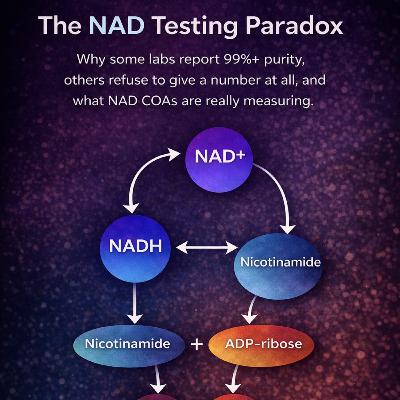
Why do some labs report 99%+ purity for NAD, while others refuse to give a purity number at all?This episode unpacks what is known as the NAD Testing Paradox.NAD is often analysed using methods designed for peptides, but chemically it behaves very differently. Because NAD naturally breaks down into related molecules like nicotinamide, ADP-ribose, AMP and ADP, purity numbers can sometimes reflect the analytical method used, rather than the actual chemical state of the molecule.Using real COA examples, this episode explores;why peptide style testing can be misleading for NADhow different labs can end up reporting very different numberscases where laboratories even disagreed on the identity of a samplewhat a properly selective NAD testing method actually looks likeThe goal isn’t to criticise labs, but to explain why NAD purity numbers can appear contradictory, and what those results may actually be measuring.
How Telehealth, US Manufacturing and 503A Ambitions are Rebuilding the Peptide MarketThe peptide market may be moving into a new phase.For years, much of the industry operated through Research Use Only (RUO) websites selling peptides in a legal grey zone. But that model is coming under pressure from payment processors, regulators, and changing mrket expectations.This episode explores what may be replacing it.Rather than a simple shift from RUO vendors to legitimate pharmacies, a more complex infrastructure appears to be emerging: telehealth platforms, physician sign-off models, compounding pharmacies, and even underground U.S. manufacturing operations trying to move toward 503A licensing over time.We break down how clinics created demand for alternative peptide supply, why telehealth has become such an attractive distribution layer, what legitimate sterile compounding actually involves, and why some operators may be using telehealth as a bridge toward a more defensible healthcare structure.The key point is that this is not a clean transition from grey market to regulated pharmacy.It is a hybrid phase in which:some operations are fully licensed pharmaciessome are Telehealth driven prescribing systemssome are clinic facing vendor networks wrapped in physician sign-offand some may be underground U.S. manufacturers trying to acquire the telehealth layer now and the 503A wrapper laterDemand for peptides hasn’t disappeared.What’s changing is the infrastructure through which that demand is served, and who manages to control the next version of the market.
For years the grey-market peptide ecosystem operated in a strange legal balance. Products were sold under “Research Use Only” labels while being widely discussed in wellness communities, clinics, and online forums. Regulators knew the contradiction existed, but the system continued to function.So what is actually destabilising the RUO model now?In this episode we look at a factor that rarely gets discussed: payment processors.Behind the scenes, vendors report increasing pressure from banks and card networks. High-risk merchant classifications, rolling reserves, frozen balances, and sudden account shutdowns are becoming common. Some companies now struggle not because they can’t produce peptides, but because they can’t reliably get paid for them.We explore how payment infrastructure is beginning to shape business behaviour across the peptide market. Vendors are experimenting with crypto checkout bridges, limiting sales volumes to avoid scrutiny, or moving toward telehealth and compounding pharmacy partnerships that qualify for healthcare merchant certifications like LegitScript.From this perspective, the shift happening in the peptide industry may not simply be RUO vendors rebranding as PUO or moving toward compounding pharmacies.It may be something more fundamental.The companies that control the rails of money may ultimately decide which peptide business models survive.
In this episode we unpack the shift from Research Use Only (RUO) peptides to the emerging Physician Use Only (PUO) label.At first glance, PUO might look like a move toward a more legitimate, physician-driven distribution model. But a closer look suggests something more complex.PUO is not an FDA regulatory category and does not automatically place peptides inside licensed pharmacy frameworks such as 503A or 503B compounding. Instead, it appears to function primarily as a liability repositioning strategy, allowing vendors to distance themselves from direct human use claims while shifting more responsibility toward physicians and clinics.We explore how the original RUO label emerged, why that model began to break down, and how some vendors are now rebranding products for “physician use” while still operating outside formal drug approval pathways.The episode also looks at real examples from companies marketing peptides to clinicians while still including research-use disclaimers , creating a hybrid system where the marketing suggests therapeutic use but the legal framework remains unchanged.The key takeaway: PUO may change the language around peptides, but it does not necessarily change the underlying regulatory structure.And while RUO vendors rebrand, a parallel system built around telehealth and compounding pharmacies may be emerging at the same time.
In this audio version episode of K-Hole Origins: My Road to Grey, I talk about my experience with a vendor known as Cora and how a seemingly normal transaction slowly turned into a lesson in how the grey market actually works.It started in March 2024. Cora approached me warmly, calling me “dear friend,” reassuring me about shipping and reship policies, and sending an Alibaba payment link for tirzepatide. The first order actually arrived, which gave me confidence to place a second, larger order for several peptides.But once testing results came back through PTDS, the picture changed. Multiple products, MOTS-C, DSIP and Epitalon, were under dosed. When I asked for a refund, the conversation shifted into what I now recognise as the classic grey-market script: reassurance, batch excuses, replacement offers instead of refunds, and endless procedural delays.The refund became a negotiation. The amount kept shrinking. I was asked to keep the agreement secret and even asked to delete posts discussing the issue. When the agreed refund still didn’t appear, the process was pushed into complicated Alibaba procedures that never actually resolved anything.In the end, the only thing that worked was a credit-card chargeback.What began with hearts, “dear friend,” and friendly messages ended with a defence letter claiming I was unreasonable and spreading rumours.
XCE goes dark, panic spreads, and the scramble to replace a dominant vendor begins. In this episode of K-Hole Origins, I share the surreal reality of early group buys, the post-XCE chaos, and the moment I realised the grey market was never just about peptides, it was about power, loyalty, and who fills the vacuum next. 
In Episode 2 of K-Hole Origins: My Road to Grey, my first peptide order from China finally arrives. What should have been a straightforward experiment quickly raises doubts and pulls me deeper into the strange dynamics of the grey-market peptide community
In this episode I share the personal story of how I entered the grey-market peptide world, from early experiences with GLP-1 medications to placing my first direct order from China, the beginning of the journey that eventually led to K-Hole.
This episode of Behind the Vial explores what happens just before peptides are freeze-dried. It looks at the stage where the peptide is still in liquid solution, covering sterile filtration, solution preparation, peristaltic pump setup, and manual vial filling under laminar airflow. Using real factory footage, the episode shows how the solution is filtered, transferred, and dispensed into vials before moving on to the lyophilisation stage.
GLP1Forum; Manipulated Labels, Word Filters & BansIn grey-market communities, information and reputation are everything.But what if the reputations people see forming online aren’t as organic as they appear?This piece explores how moderation tools, word filters, bans, and platform design can influence which narratives survive and which disappear.Sometimes rumours spread.Sometimes they’re built directly into the system.
This episode explains the lyophilisation process used in peptide manufacturing, showing how liquid formulations are freeze dried to improve stability. It covers the key stages of freezing, primary drying, secondary drying, and final stoppering, and explains how water is removed by sublimation under vacuum. Using real factory footage from Aaron’s Biochemapi facility, the episode walks through what happens inside the freeze dryer and shows how partially stoppered vials become sealed, freeze-dried products.
This is a summary of my interview with Alex, co-founder of Uzorak, a new peptide testing lab based in Zagreb, Croatia, planning to operate globally.Alex comes from a health-tech background, including a DNA analytics company , and says his own peptide research is what pushed him into building a testing lab. His core argument is simple: in an unregulated market, testing standards should be higher, not lower. He believes people are going to use these compounds regardless, so harm reduction starts with better information about what’s actually in the vial.Uzorak’s offering goes beyond the standard “HPLC-UV purity + mass confirmation” COA. Their “Advanced LCMS Peptide Analysis” is positioned as a bundled package: high resolution LC-MS, impurity identification and quantification, MS/MS sequence confirmation, and tandem MS fingerprinting to check for structural damage versus a reference. They also add a scoring model with published weights, purity, potency, and integrity, plus risk triggers, and they separate “alteration” signals like oxidation and deamidation from “hazard” categories like hydrolysis, cleavage, and synthesis residues.One of the big clarifications in the interview is language: “structure” here doesn’t mean 3D protein folding. Alex notes that folding becomes relevant for large biologics like interferon, filgrastim, or erythropoietin, but for GLP-class peptides, they’re focused on identity, sequence integrity, and detectable chemical modification.We also talked about thresholds and risk messaging. Uzorak presents specific warning triggers, and Alex described red-level findings as potentially more serious, while also acknowledging that analytical flags are not the same thing as clinical diagnosis. They are not ISO/IEC 17025 accredited yet, but say they follow many of the principles where applicable. Sterility, endotoxin, and heavy metals aren’t included by default currently, though an ICH Q3D-aligned heavy metals panel is planned.Bottom line: this interview focuses on analytical substance, not surface - level claims. It’s about whether grey market testing evolves beyond a reassuring purity percentage into a more complete picture , what’s in the peak, what’s outside it, what’s degraded, and what the limits of testing still are.www.uzorak.com
In this deep dive, we break down drop-shipping in grey peptide markets and why it’s not “just logistics.” In this world, logistics determines custody, accountability, and verification. A polished storefront that accepts PayPal or credit cards may actually be acting as a broker: taking fiat payments, converting to crypto, and routing orders to an upstream supplier the buyer never sees.We also unpack how group buys can function as drop-shipping infrastructure, including direct ship models where organizers never physically handle inventory, and “reverse drop-ship” setups where resellers route customer orders through a group buy to avoid holding stock.Then we connect the structure to the biggest practical failure point: COAs. In layered supply chains, COAs can become representative rather than batch specific, outdated, or disconnected from what a customer actually receives, especially when vials lack unique identifiers.The key mindset shift: stop asking “is this vendor legit?” and start asking “who controls the batch?” Because in a drop-ship model, supply can change overnight, and buyers are often the last to know.Bottom line: drop-shipping isn’t automatically evil. The risk comes from opaque structure, where trust is built on interface and reassurance rather than traceability.
If you’ve ever opened a COA, seen “99% purity,” and felt instant relief… this one’s for you. Because that number is usually HPLC-UV purity, and UV purity isn’t a statement about the whole vial, or even a guarantee of molecular identity. It’s a statement about what the detector can see under a specific method, and how the lab calculates the percentage.In regulated pharma, that UV number sits inside a full GMP quality system, validated processes, controlled inputs, stability programs, and strict release limits. In the grey market, you don’t have that surrounding framework. So the exact same “99%” can be far less reassuring.We break down what UV purity actually measures, why it can miss co-eluting variants and non-UV-friendly contaminants, and why “high purity” doesn’t automatically equal “high net peptide content” once salts, water, and unexpected add-ins enter the picture. We also touch on why some risks, like heavy metals, sterility, and endotoxin, require completely different tests.Bottom line: HPLC-UV isn’t useless. But in the grey market, UV alone can’t carry the meaning people want it to carry.
Same Script, New NameJust when the Bella story looked finished, the pattern resurfaced, under a new name.In Episode 5, the script repeats. A new vendor appears. A polished pitch. A confident backstory. Claims of factory access, quality control, and “no pressure.” The language feels familiar. The structure feels familiar. And then the price list lands, almost identical in layout, formatting, and product order to the ones we’ve already seen.Although the branding is cleaner., the spelling errors are still there. The presentation is sharper. But the workflow hasn’t changed.When questioned, the urgency increases. Off platform contact details appear early. Reassurance replaces proof. Pressure stays polite but persistent. The credibility build follows the same ladder: documentation, testimonials, confidence, deflection.And when scrutiny grows, so does the performance.Episode 5 explores what happens when a scam doesn’t collapse, it evolves. New name. New profile picture. Same funnel.
Undercover at CheckoutThis episode follows an undercover buyer taking Bella to the point that matters: payment.The pattern stays consistent: hook with cheap shipping + fast delivery, deflect COA questions, then pivot the conversation off Discord and into WhatsApp when risk questions appear.At checkout, the “payment ladder” shows up: push crypto first, fall back to PayPal Friends & Family, then rotate to new PayPal accounts (“cashier,” “sister”) when the buyer hesitates. Under pressure, Bella leans on the Zen COAs again, not as proof, but as a reassurance patch, until scrutiny leads to blocking.The typo might change. The workflow doesn’t.
Touchdowns and Testimonials, when the threads convergedThe morning after the Bella interview, Plotting Profits reappears with the same sales loop behaviour: a casual re entry (“hello”), a guilt nudge (“you didn’t get back to me”), then the hook (“prices went down recently”) followed by the familiar move: get the buyer off platform and keep the catalogue moving.That trail leads straight into Alisa’s Discord, branded as “science-based discussion,” but structured as the opposite: broadcast only, read only, no real community conversation. The “discussion” is a controlled feed, dominated by product posts, packaging shots, and carefully placed content meant to look like legitimacy.Then come the touchdowns and testimonials, the emotional, over the top praise (“God sent… only person who stood by me…”) that reads less like normal customer feedback and more like manufactured trust. Alisa replies warmly, then drops the sales line in the same breath: “Still taking orders family.” Gratitude → bonding → transaction.And then the paperwork returns.When you ask about payment and shipping, the setup feels familiar. When you ask about documentation, Zen appears again, the same name that keeps resurfacing across sellers. Explanations shift depending on pressure, but the pattern stays consistent: keep the paperwork sounding official without locking down who it actually belongs to.Episode 3 is where the series title earns its keep: the story isn’t held together by one lie. It’s held together by repetition, the same scripts, the same credibility props, the same “proof,” reused under different faces.