Discover Paper Talk
Paper Talk

693 Episodes
Reverse
Researchers have developed a novel engineering strategy to overcome the common tendency of lipid nanoparticles (LNPs) to accumulate in the liver rather than other organs. By utilizing passive targeting through abdominal injection, the study found that larger particles—roughly 300 nanometres in diameter—selectively infiltrate the pancreas due to its uniquely thin protective capsule. To ensure effective mRNA expression, scientists used smaller lipids that expand upon contact with bodily fluids to form a specific protein corona. This protein layer is enriched with lipid-binding molecules that interact with VLDL receptors, allowing for the precise delivery of therapies to mesenchymal stromal cells within the pancreas. This breakthrough moves beyond trial-and-error methods toward a knowledge-driven design capable of treating aggressive pancreatic cancers and other previously unreachable diseases. The successful application in animal models provides a blueprint for customizing nanoparticles to target specific tissues based on their unique biological and anatomical environments.References: https://www.nature.com/articles/d41586-026-00294-5
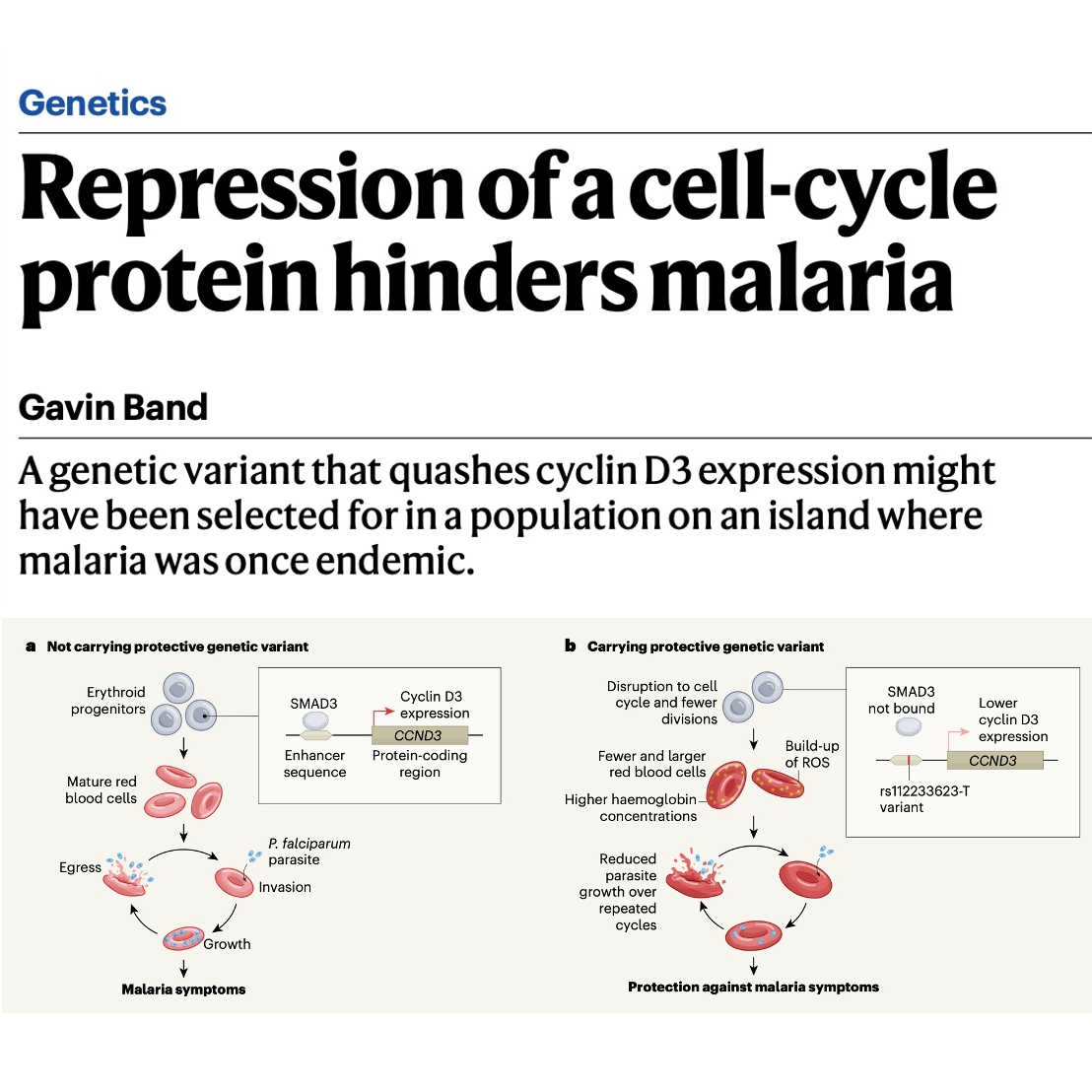
Researchers have identified a genetic variant called rs112233623-T that appears to provide natural protection against malaria by reducing the expression of the cyclin D3 protein. This specific mutation, which is unusually prevalent in the Sardinian population, alters how red blood cells develop, resulting in cells that are fewer in number and larger in size. These modified blood cells contain higher levels of reactive oxygen species, creating an internal environment that hinders the growth of the Plasmodium falciparum parasite. Because Sardinia was historically a region with high malaria transmission, scientists believe this trait was favored by natural selection to help inhabitants survive the disease. The discovery of this biological pathway is significant because it suggests that existing cancer medications targeting similar cell-cycle proteins could potentially be adapted for malaria treatment.References: https://www.nature.com/articles/d41586-026-00289-2
Researchers have developed Foli-seq, a new noninvasive sequencing method that captures host messenger RNA from cells naturally shed into stool. This technique overcomes historical obstacles like RNA degradation and high bacterial interference to provide a detailed map of intestinal and immune function. Testing in murine colitis models demonstrated that the tool can track epithelial damage and recovery while identifying specific biomarkers that predict disease severity. By combining this data with microbiome profiling, the authors uncovered complex host–microbe interaction networks that were previously difficult to observe. Finally, the study successfully applied Foli-seq to human IBD patients, stratifying them into subgroups that correlate with clinical symptoms and inflammatory activity. This approach offers a powerful, longitudinal alternative to invasive biopsies for monitoring chronic gastrointestinal diseases.References: Huang Y, Sun Y, Ronda C, et al. Fecal exfoliome sequencing captures immune dynamics of the healthy and inflamed gut[J]. Nature Biotechnology, 2025: 1-11.
This research investigates the biological paradox of how viruses produce proteins efficiently despite using genetic codes that are poorly adapted to human host cells. Typically, human cells use mRNA circularization to monitor and slow the translation of sequences with rare, non-optimal codons. The study reveals that viral 5′ untranslated regions (UTRs) actively block this looping process, allowing viral RNA to bypass the host's natural quality control mechanisms. By remaining linear, these viral transcripts achieve high-speed protein synthesis regardless of their codon quality. These findings offer a new explanation for the virulence of pathogens like SARS-CoV-2 and suggest novel strategies for improving vaccine design and antiviral therapies. This discovery identifies mRNA shape as a primary regulator of how genetic instructions are converted into functional proteins.References: Liu H, Liu Y. How viral RNAs escape a host mechanism that controls translation[J]. Mol. Syst. Biol, 2009, 5: 311.
This article explores the integration of untargeted mass spectrometry (MS) and wearable biosensors to advance noninvasive health monitoring. While MS serves as a powerful discovery engine for identifying complex biomolecular patterns in fluids like sweat and saliva, wearable devices provide the continuous, real-time data necessary for tracking dynamic physiological changes. The authors establish specific criteria for translating laboratory-discovered biomarkers into on-body applications, emphasizing the need for molecular stability and high sensitivity. By bridging these two technologies, the researchers propose a move away from episodic clinical testing toward a personalized, longitudinal monitoring framework. This synergistic approach aims to transform how we detect and manage conditions ranging from metabolic stress to chronic disease in real-world settings.References: Kim M J, Lasalde-Ramírez J A, Heng W, et al. Biomolecular profiling for noninvasive health monitoring[J]. Nature Biotechnology, 2026: 1-14.
Researchers have developed a novel neuroimmunotherapy for Alzheimer’s disease by engineering astrocytes to express chimeric antigen receptors (CARs). While traditional treatments rely on antibodies that often require high doses and repeated administration, this CAR-A therapy enables a one-time, noninvasive delivery to transform brain cells into "super-phagocytes." These specialized cells are designed to specifically target and clear amyloid-β aggregates, which are primary drivers of neurodegeneration. In mouse models, a single treatment significantly reduced existing plaque burden and effectively prevented new amyloid accumulation while decreasing damage to surrounding neurons. The study also found that these engineered astrocytes communicate with microglia, shifting the brain's immune environment toward a more protective, homeostatic state. Ultimately, this platform offers a customizable and scalable strategy for treating complex neurodegenerative conditions beyond typical oncology applications.References: Chen Y, Liu Y, Nguyen K, et al. Targeting amyloid-β pathology by chimeric antigen receptor astrocyte (CAR-A) therapy[J]. Science, 2026, 391(6789): eads3972.
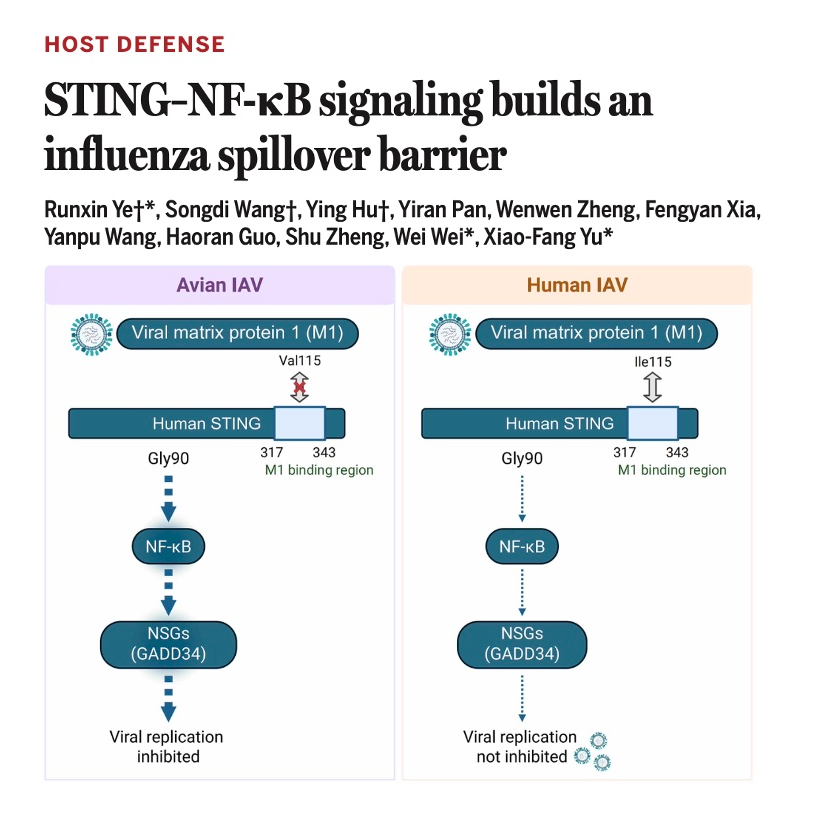
This research identifies the STING–NF-κB–GADD34 signaling pathway as a critical innate immune barrier that restricts the transmission of avian influenza A viruses into humans. While the STING protein is well-known for triggering interferons, this study reveals that it also activates an IRF3-independent antiviral response through a specific Gly90 residue. This activation induces the expression of GADD34, which directly binds to and inhibits the viral polymerase complex to block replication. To counter this defense, human-adapted influenza strains have evolved a specific isoleucine mutation at position 115 of the M1 protein. This mutation allows the virus to evade human immune detection, whereas avian strains lacking this adaptation remain restricted. Consequently, monitoring the M1-115 variant provides a vital molecular marker for predicting the spillover risk of emerging pandemic threats.References: Ye R, Wang S, Hu Y, et al. STING–NF-κB signaling builds an influenza spillover barrier[J]. Science, 2026, 391(6788): eads4405.
This research identifies a negative feedback loop between two proteins, LEAFY (LFY) and TERMINAL FLOWER1 (TFL1), which regulates the growth patterns of plant shoots. While the florigen protein (FT) typically signals plants to stop growing and start producing flowers, the inflorescence meristem at the tip of the shoot remains indeterminate, allowing for continuous growth. The study reveals that LFY activates TFL1 specifically in this central growth zone, while TFL1 simultaneously restricts LFY levels to prevent the tip from turning into a flower. Using computational modeling and genetic experiments in Arabidopsis, the authors demonstrate that this internal balancing act buffers the plant against environmental fluctuations. This mechanism ensures that while lateral meristems transition into flowers for reproduction, the main shoot maintains its stem cell pool for sustained development.References: Huang T, Hodgens C, Prakash S, et al. A negative feedback loop between TERMINAL FLOWER1 and LEAFY protects inflorescence indeterminacy[J]. Science, 2026: eadv5429.
This research article identifies the structural basis for how the protein kinase mTORC2 selectively recruits and phosphorylates Akt and related AGC family kinases. Researchers utilized expressed protein ligation to create semisynthetic Akt probes, allowing them to trap the transient kinase-substrate interaction for high-resolution cryo-electron microscopy analysis. The study reveals that mTORC2 recognition depends on two distinct long-range docking interfaces—the CRIM domain and the "N-mooring" of the mSin1 subunit—rather than the local amino acid sequence surrounding the phosphorylation site. These findings demonstrate that mTORC2 directly phosphorylates both the Ser473 hydrophobic motif and the Thr450 turn motif through a coordinated conformational change at the plasma membrane. By defining these specific binding surfaces, the authors provide a molecular framework for developing therapeutic inhibitors that selectively target mTORC2 over the closely related mTORC1 complex. This work ultimately clarifies long-standing questions regarding substrate specificity in vital signaling networks associated with cancer and diabetes.References: Taylor M S, Chen M, Hancock M, et al. Structural basis for the recruitment and selective phosphorylation of Akt by mTORC2[J]. Science, 2025: eadv7111.
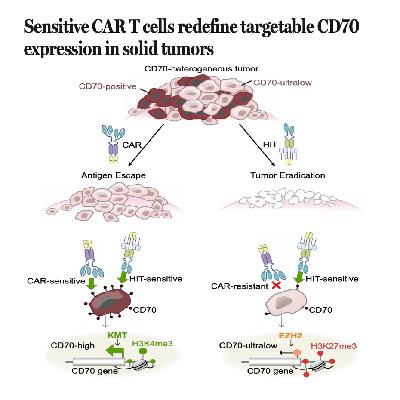
This paper describe a breakthrough in cancer immunotherapy addressing why CAR T cells often fail to eliminate solid tumors. Research reveals that many tumors appearing to be CD70-negative actually possess ultralow levels of the antigen that are hidden by epigenetic silencing via the protein EZH2. To overcome this, scientists engineered a more sensitive HLA-independent T cell (HIT) receptor capable of detecting these trace amounts of CD70. When combined with specific costimulatory ligands, these HIT T cells successfully achieved complete tumor eradication in models of kidney, ovarian, and pancreatic cancer. This discovery suggests that CD70 is a universal target across many malignancies if treated with high-sensitivity receptors. Ultimately, the study demonstrates that antigen heterogeneity is often a matter of detection limits rather than a total absence of the target.References: Hanina S A, Park T, Lopez M, et al. Sensitive CAR T cells redefine targetable CD70 expression in solid tumors[J]. Science, 2026, 391(6788): 896-905.
This research explores how cells terminate the integrated stress response (ISR) to resume normal protein synthesis. The study identifies that the protein R15B plays a critical role by facilitating the dephosphorylation of P-eIF2 while it is still attached to the translation factor eIF2B. Previously, it was unclear how this process occurred because the inhibited complex appeared structurally inaccessible to traditional repair mechanisms. Using advanced cryo-electron microscopy, the researchers demonstrated that R15B effectively "unlocks" the inhibited eIF2B, allowing it to become active again. This discovery provides a vital link in understanding cellular recovery, as failing to end the stress response can lead to cell death. Ultimately, the findings highlight a sophisticated molecular rescue operation that maintains cellular homeostasis and fitness.References: De Miguel C, Thorkelsson S R, Fatalska A, et al. Termination of the integrated stress response[J]. Science, 2025: eadw5137.
This research article presents a massive single-cell chromatin accessibility atlas that maps the biological process of aging across 21 different mouse tissues. Using an advanced sequencing technique called EasySci-ATAC, the authors analyzed over 10 million nuclei to observe how the physical structure of DNA changes as an organism grows older. The study identifies significant remodeling of the epigenomic landscape, revealing that aging triggers both tissue-specific shifts and universal patterns of cellular depletion or inflammation. Furthermore, the data highlights sexual dimorphism, showing that male and female cells often follow distinct molecular trajectories during the aging process. By providing this comprehensive resource, the researchers aim to offer a foundational roadmap for developing targeted therapies to restore youthful tissue function and combat age-related diseases.References: Lu Z, Zhang Z, Xu Z, et al. Organism-wide cellular dynamics and epigenomic remodeling in mammalian aging[J]. Science, 2026, 391(6788): eadw6273.
This research study investigates how learning a task transforms the way neurons in the macaque visual cortex process and share information. Contrary to the classic belief that the brain improves efficiency by reducing overlapping signals, the authors discovered that information redundancy actually increases as animals become more proficient at a task. By recording neural activity over several weeks, they observed that neurons began to share more task-relevant data through feedback and internal inference rather than operating as independent sensors. This shift suggests that sensory processing is a bidirectional inference process where prior expectations and incoming evidence are integrated across the neural population. Ultimately, the study reveals that the brain prioritizes distributing information across many neurons to support robust decision-making rather than simply eliminating noise or redundancy.References: Liu S, Pletenev A, Haefner R M, et al. Task learning increases information redundancy of neural responses in macaque visual cortex[J]. Science, 2026, 391(6789): 1029-1035.
Researchers have developed a comprehensive single-cell atlas of the mouse brain to map how neural activity changes across the 24-hour circadian cycle. By using advanced tissue clearing and 3D imaging techniques on 144 brains, the study identified that nearly 80% of brain regions exhibit significant rhythmic patterns under constant darkness. The data reveals that while many areas peak during the animal's active night phase, others, such as those governing sleep and vision, activate during the day. This research highlights the spatiotemporal coordination of the entire brain, showing how different subregions specialize their activity based on time. To support future neuroscience and pharmacological studies, the authors provided an open-access database for exploring these global rhythms. This resource establishes a vital temporal framework for understanding both healthy brain function and the dynamics of neurological diseases.References: Yamashita K, Kinoshita F L, Yoshida S Y, et al. A whole-brain single-cell atlas of circadian neural activity in mice[J]. Science, 2025: eaea3381.
This article identifies a self-perpetuating tumor-immune-neural circuit that drives cancer cachexia, a debilitating syndrome of muscle and fat wasting. The study reveals that tumors secrete CSF1, which triggers macrophages to produce the hormone GDF15. This hormone acts on the brainstem to activate the sympathetic nervous system, which in turn releases norepinephrine that further stimulates tumor cells to produce more CSF1 via a ZIP4-ZFP64 signaling pathway. This feedback loop disrupts systemic energy balance, leading to anorexia and severe tissue loss in pancreatic, lung, and skin cancers. Experimental results show that pharmacologically blocking this axis with anti-GDF15, anti-CSF1R, or RET inhibitors effectively reverses wasting symptoms. Consequently, targeting this tripartite interaction offers a promising therapeutic strategy for improving the quality of life and survival of patients with advanced cancer.References: Shi X, Arreola A X, Zhou Z, et al. Tumor-immune-neural circuit disrupts energy homeostasis in cancer cachexia[J]. Cancer cell, 2026.
This research introduces a novel mathematical model to examine how ecological relationships within the human gut microbiome change during illness. While traditional methods focus on microbial diversity, which can be inconsistent across different conditions, these findings suggest that dysbiosis is fundamentally defined by a shift in interaction types. Healthy gut environments are typically characterized by competition for resources, whereas diseased states are dominated by cooperative cross-feeding among specific bacterial clusters. To quantify this transition, the authors developed the Ecological Network Balance Index (ENBI), a diagnostic metric that distinguishes between healthy and pathological states across various diseases like IBD and colorectal cancer. The study demonstrates that this index not only identifies the presence of disease but also effectively tracks its clinical progression. Ultimately, this framework provides a more mechanistic understanding of microbial stability and offers a robust, non-invasive tool for early disease detection.References: Corral López R, Bonachela J A, Dominguez-Bello M G, et al. Imbalance in gut microbial interactions as a marker of health and disease[J]. Science, 2026, 391(6788): 890-895.
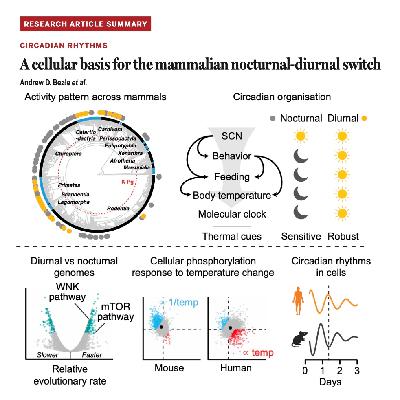
This research identifies a cell-intrinsic, thermodynamic mechanism that governs the biological switch between nocturnal and diurnal activity in mammals. While the brain's master clock remains the same across species, peripheral cellular clocks in diurnal mammals have evolved to respond differently to daily cycles of temperature and osmolality. Specifically, diurnal cells are more robust and less sensitive to these environmental perturbations due to accelerated evolution in the mTOR and WNK signaling pathways. In contrast, nocturnal cells show a high level of sensitivity, with biochemical processes like protein synthesis accelerating in response to heat. The study demonstrates that manipulating mTOR activity can effectively shift the circadian behavior of nocturnal cells to a more diurnal-like pattern. These findings suggest that temporal niche selection is rooted in fundamental genetic and biochemical adaptations to cellular thermodynamics.References: Beale A D, Christmas M J, Rzechorzek N M, et al. A cellular basis for the mammalian nocturnal-diurnal switch[J]. Science, 2026, 391(6788): eady2822.
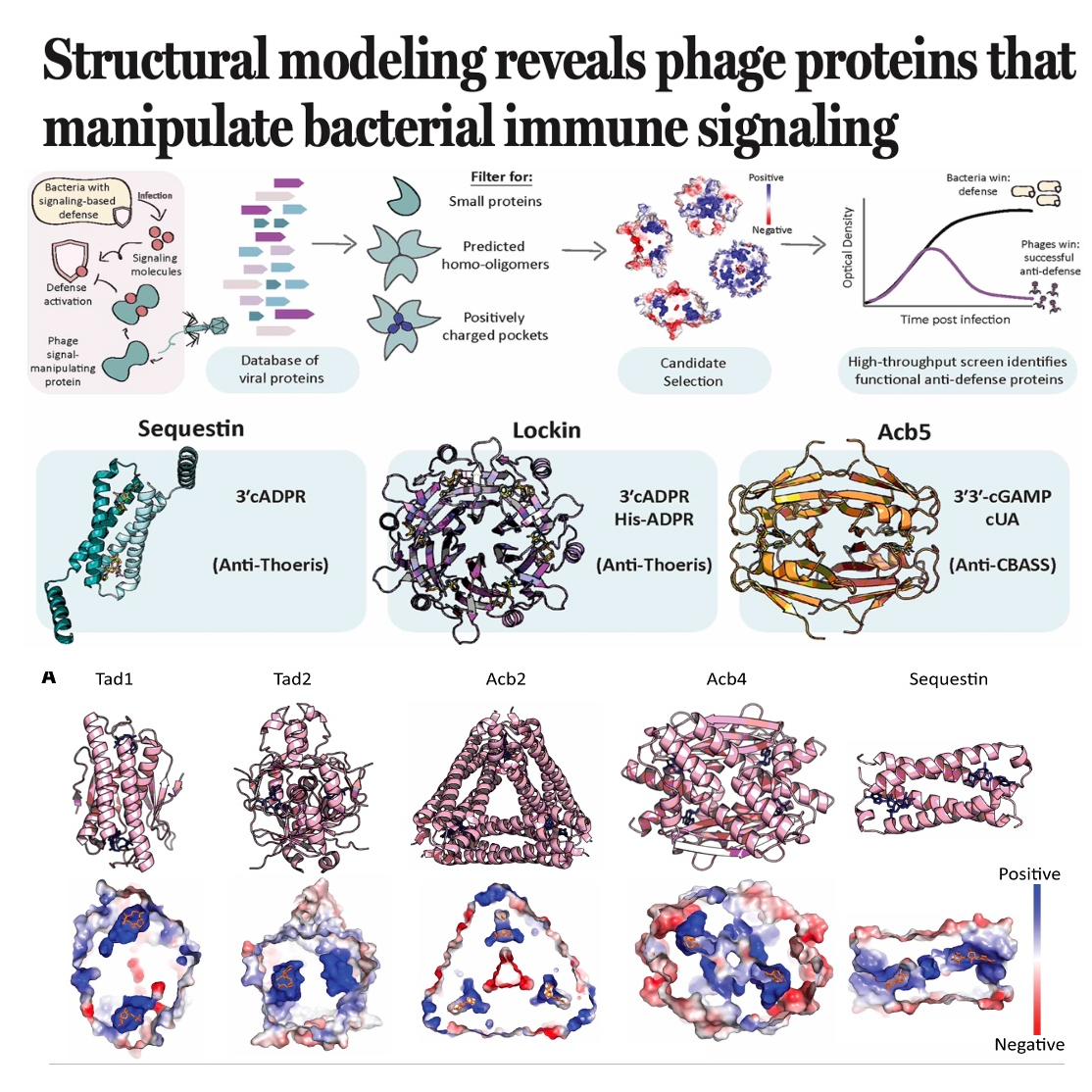
This research identifies and validates new families of phage-encoded antidefense proteins that allow viruses to bypass bacterial innate immunity. By analyzing common structural traits, researchers developed a computational pipeline to discover viral proteins that either sequester or enzymatically degrade nucleotide signaling molecules. This approach led to the identification of Sequestin and Lockin, two "sponge" protein families that neutralize the Thoeris defense system, and Acb5, an enzyme that disables CBASS immunity. Structural modeling and biochemical assays confirmed that these proteins are widespread in nature, appearing in thousands of viral genomes including the well-studied T4 phage. The study demonstrates that structure-guided discovery is a powerful method for uncovering the diverse strategies viruses use to subvert host immune signaling across different domains of life.References: Tal N, Hadary R, Chang R B, et al. Structural modeling reveals phage proteins that manipulate bacterial immune signaling[J]. Science, 2026, 391(6789): eaea1761.
Scientists have successfully developed a method to reconstitute the testicular environment using only mouse pluripotent stem cells, bypassing the need for embryonic tissue. By manipulating specific signaling pathways, researchers guided these stem cells through a bipotential gonadal state into functional testicular somatic cell-like cells (TesLCs). When combined with primordial germ cell-like cells, these induced tissues formed testicular organoids that supported the development of spermatogonial stem cells. These laboratory-grown stem cells were capable of producing mature sperm after transplantation into mice, eventually leading to the birth of healthy, fertile offspring. The study also highlights a fundamental asymmetry in sex determination, demonstrating that while male differentiation strictly requires a Y chromosome, male cells can still be induced to support female egg development. This breakthrough provides a sophisticated platform for studying reproductive biology and advancing in vitro gametogenesis across various species.References: Yoshino T, Sasada H, Sato T, et al. Reconstitution of sex determination and the testicular niche using mouse pluripotent stem cells[J]. Science, 2026, 391(6788): eaea0296.
The paper introduces FlashS, a novel computational framework designed to identify spatially variable genes (SVGs) within massive spatial transcriptomics datasets. Current methods often struggle to balance statistical accuracy with the computational scalability required for million-cell atlases, frequently failing due to high memory demands or simplified models. FlashS overcomes these limitations by transforming spatial testing into the frequency domain using Random Fourier Features, which allows for the detection of complex, multi-scale patterns without the need for expensive distance matrices. The method incorporates a three-part test to handle extreme zero-inflation and a kurtosis-corrected null distribution to ensure precise statistical calibration. Across diverse benchmarks and biological tissues like the human heart and mouse brain, FlashS consistently outperforms existing tools in both speed and the recovery of biologically meaningful gene programs. Consequently, it offers a robust, memory-efficient solution for researchers mapping the functional organization of complex tissues at an unprecedented scale.References: Yang C, Zhang X, Chen J. Frequency-domain kernels enable atlas-scale detection of spatially variable genes[J]. bioRxiv, 2026: 2026.03. 12.711372.